Technical Support技术支持
CONTACT US
 400 179 0116
400 179 0116
24-hour service hotline marketing@ldraft.comE-mail
marketing@ldraft.comE-mail
Isolation and Culture of B Cells
source:QiDa technoligy views:316 time:2026-03-31
B cells play a central role in immune development. They initially enter the bloodstream from the bone marrow as naive B cells, which can migrate to lymphoid tissues such as the spleen, lymph nodes, and tonsils for further development. Some naive B cells migrate to lymphoid follicles, where germinal center B cells can differentiate into memory B cells and plasmablasts (PBs)/plasma cells (PCs). Although most PBs/PCs eventually enter the bloodstream, a small number ultimately reside in the bone marrow and undergo terminal differentiation into long-lived plasma cells.
Let's start with the extraction of B cells:
Collect 2 ml of venous blood into a test tube containing heparin solution (50 μg/ml blood sample), mix well to anticoagulate the blood, and dilute with PBS at a ratio of 1:1.
Pipette 2 ml of lymphocyte separation medium (Qida Biotech, Cat. No.: SD0059) into a graduated centrifuge tube. Tilt the tube at 45°, and slowly add the diluted whole blood onto the separation medium along the tube wall using a capillary pipette, taking care to maintain a clear interface between the two layers.
Centrifuge at 1500 r/min for 20 min at 18–20 ℃ using a horizontal centrifuge.
Carefully remove the centrifuge tube and slowly aspirate the lymphocytes in the second layer with a 1 ml pipette. Balance the tubes and centrifuge again. If red blood cells remain, wash an additional time with PBS.
Discard the supernatant, and resuspend the pelleted cells in culture medium for subsequent use.
B Cell Purification
(a) Transfer 1 mg of carboxyl magnetic beads (approx. 100 μl bead suspension; mix thoroughly by ultrasonication before use) into a 1.5 ml Eppendorf tube, and vortex for 5–10 min.
(b) Take an appropriate volume of bead suspension, resuspend the cells in 20 volumes of PBS + 0.2% BSA solution, then place on a magnetic stand for 5 min until the beads are fully adsorbed.
Note: The ratio of magnetic beads to mononuclear cells or whole blood is 0.5 mg beads per 1×10⁷ mononuclear cells or per 1 ml whole blood.
(c) Carefully aspirate the supernatant with a pipette to remove the secondary antibody detached from the beads during storage. Resuspend the beads in 100 μl of PBS + 0.2% BSA solution. Add the primary antibody at a ratio of 5 μg primary antibody per 1 mg magnetic beads, mix well, and incubate for 30 min at room temperature on a sample mixer for activation.
(d) Add 2 ml of PBS + 0.2% BSA solution, place on a magnetic stand for 5 min, then aspirate the supernatant to remove unbound primary antibody. Repeat this step once. Resuspend the beads in 100 μl of PBS + 0.2% BSA solution.
(e) Adjust the concentration of cells to be separated to 1×10⁷ cells/ml using complete B cell medium (Qida Biotech, Cat. No.: P3901). Add the cell suspension to the bead mixture, incubate at room temperature for 15 min with gentle mixing once halfway, then place on a magnetic stand for 10 min. Aspirate the supernatant.
(f) Resuspend the bead-bound cells in complete B cell medium and use directly for culture.
Alright, check the figure below — B cell isolation was successful!
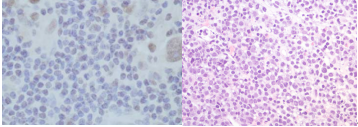
Day 1 of B cell culture Day 7 of B cell culture







